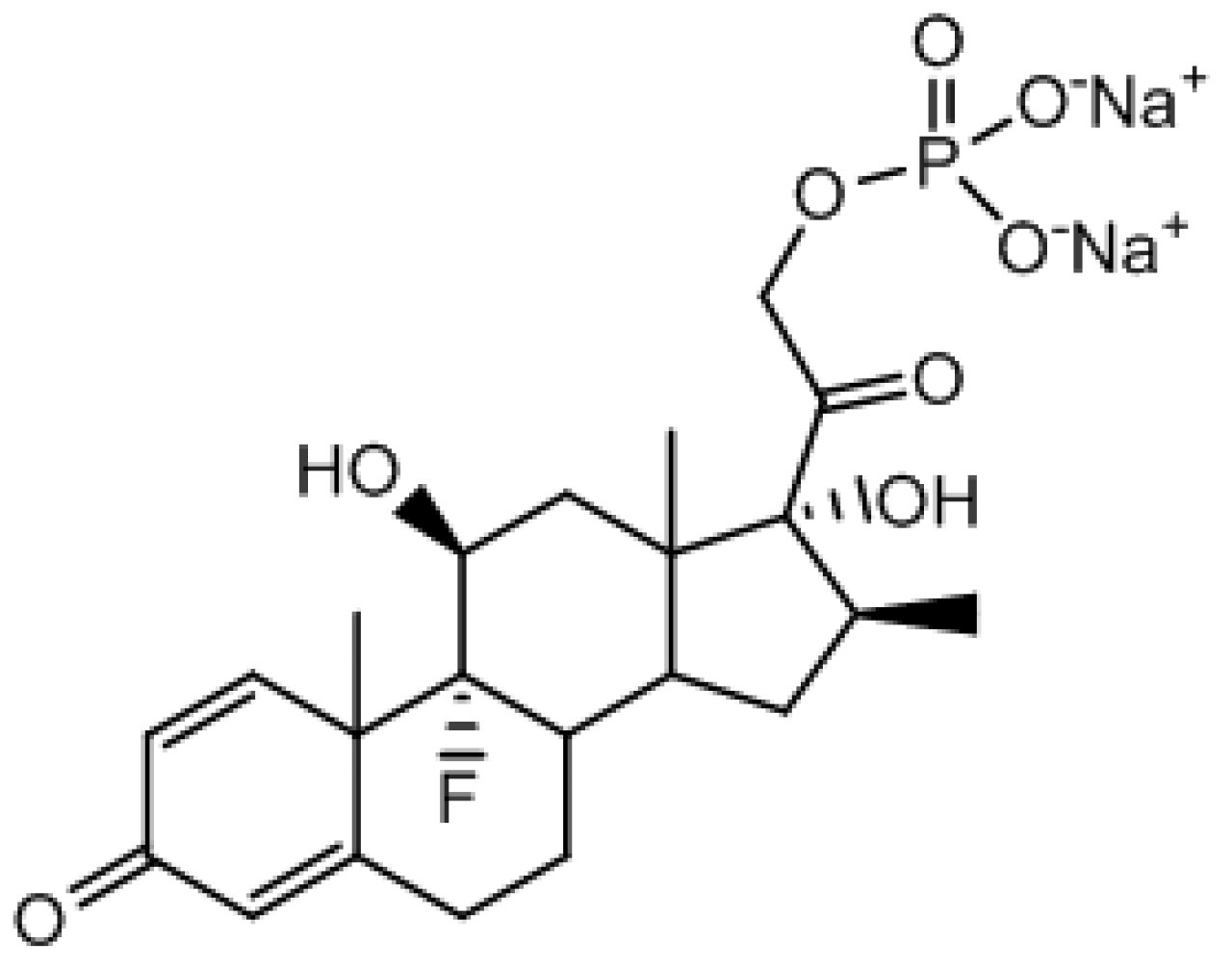
Betamethasone Disodium Phosphate
API Product
Product Status:
Commercial
Regulatory Status:
CA DMF
JP DMF
US DMF
Production Sites:
- Hovione Loures
Product Type:
- Corticosteroids
CAS Number:
151-73-5
Modes of Application:
- Topical
- Ophthalmic
- Injectable
Common Indications:
- Allergic reactions
- Ophthalmic conditions
Last Inspection:
FDA May 2018
Hovione produces Betamethasone Sodium Phophate since 1980 and has over 50 years experience in the manufacture of corticosteroids.
Injectable quality available on demand from the European producer with an unblemished Regulatory track record.
Approved in branded and generic human applications.
This is not to be construed as a representation of non-infringement or as an offer to sell in those countries where such would constitute an infringement of third parties' patent rights.
Highlight
Contact Us
Please contact us if you have inquiries about our offering
https://go.hovione.com/l/47122/2014-08-06/9grc